ELECTRONIC Health RECORDS
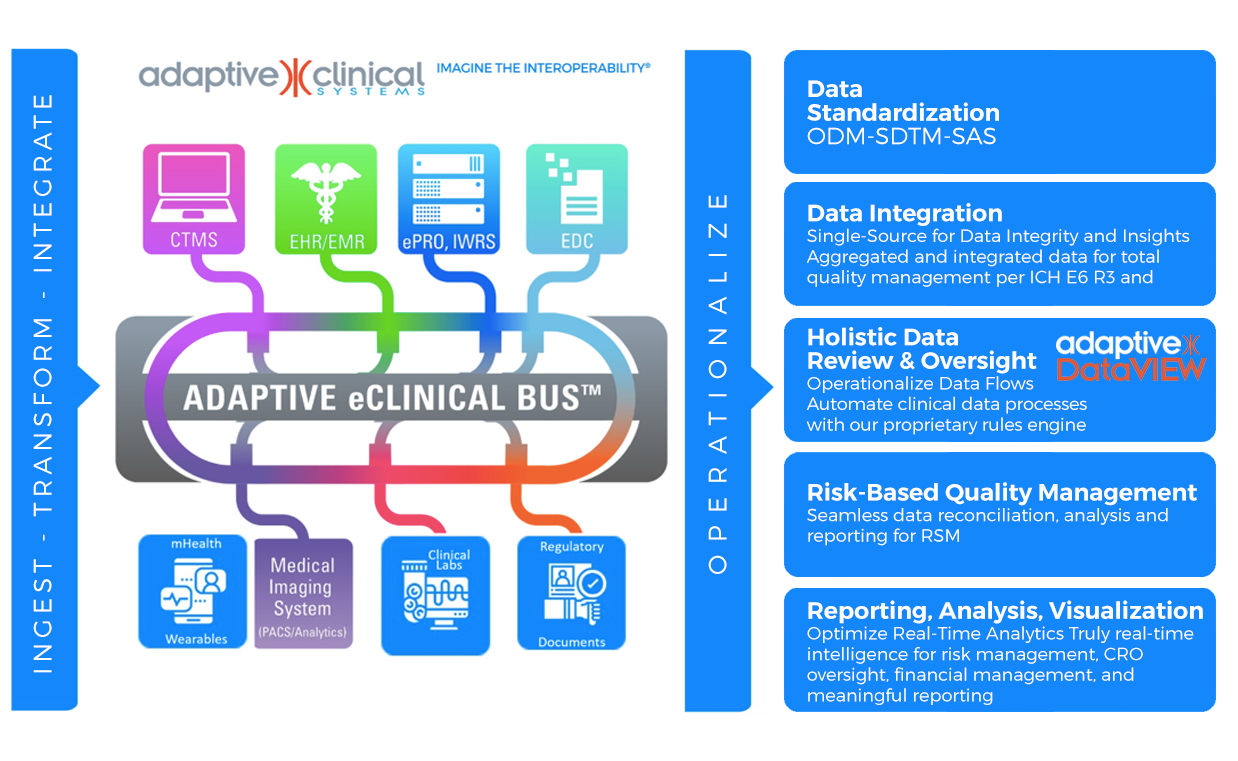
Quickly and efficiently incorporate Electronic Health Records and
Electronic Medical Records data using the Adaptive eClinical Bus®
The Adaptive eClinical Bus offers connectivity to nearly all Public Health Information found in electronic health records and electronic medical records managed by Epic Systems Corporation and Cerner Corporation.
Sponsors and CROs can quickly add data streams for any EHR. Whether your challenge is biometric data, EMR to analytics or retrospective studies, Adaptive Clinical Systems has connections to some 30+ EMR systems. Ask for a demo today.

Alis

Allegiance MD

AmazingCharts

Canvas

CareCloud

Cerbo


CharmHealth

Dr Chrono


Elation

EyeMD EMR

Greenway


MD Logic

MedHost

Meditech



Next Gen

Office Practicum

Open Dental


Praxis

Quickly identify subjects that meet study inclusion/exclusion criteria

Directly import demographic information
Easily map routine visit data into eCRF’s
Adaptive Clinical notes a number of efficiencies and core benefits for integrating EMR/EHR into EDC systems that include:
-
Reduce the complexity and cost of data transcription services to the eCRF or the paper case report form
-
Eliminate the need for second source-data verification
-
Eliminate transcription errors and access more accurate data
-
Reduce investigator and site burden by streamlining the need for multiple EDC systems
-
Integrate data efficiently from multiple healthcare clinical information systems (e.g., radiology information systems, laboratory information systems).
-
Speed stakeholder access to data (Data Science, Clinical Operations, Sites/Patients)
-
Reduce patient burden and improve provider satisfaction
For more information on the benefits of adding EHR/EMR for your clinical trials, please see our brochure.
Gain More Insights with EHR/EMR Data
When Sponsors and CROs capture data from electronic medical records (EMR) and electronic health records (EHR) it’s a win-win – for sponsors, CROs and patients. Not only does it reduce costs and improve efficiencies in clinical trials, it adds a new dimension to the patient profile and allows both health professionals treating patients as well as those overseeing clinical trials to have a more complete picture of the patient’s health. The drive to reduce the cost and complexity of trials is leading to a heightened awareness for a better understanding of patient conditions, more specific therapeutic targeting, improved use of EMRs and electronic technologies for identification of patients and ascertainment of events. EHR may include a patient’s medical history, diagnoses, treatment plans, immunization dates, allergies, radiology images, pharmacy records, and laboratory and test results.
Create Bi-Directional Workflows
As you look to incorporate EMR data into decentralized trials, Adaptive Clinical’s platform facilitates bi-directional data workflows across all study partners and eClinical data sources. The FDA1 defines interoperability as “the ability of two or more systems or components to exchange information and to use the information that has been exchanged.” The FDA also notes that there are benefits to clinical investigations, patients and other health care providers when interoperability and automated electronic exchange of information exists between the EHR and the sponsor’s electronic systems. The interoperability between EHR and EDC systems may simplify data collection for a clinical investigation by enabling clinical investigators and study personnel to capture source data at the time of a patient’s point-of-care visit.
Leverage Adaptive Clinical’s Interoperability Solutions
The Adaptive eClinical Bus® solution integrates clinical study data from multiple systems and platforms — EDC, eCOA, CTMS, Medical Imaging, IRT, wearables and analytics/data visualization systems. By incorporating Adaptive DataVIEWTM, sponsors and CROs will obtain a comprehensive data visualization solution that complements and extends any data source from the Adaptive eClinical Bus® to operationalize, gain insight from, exchange and visualize data more quickly. For example, data elements originating in an EHR (e.g., demographics, vital signs, past medical history, past surgical history, social history, medications, adverse reactions) can automatically populate the eCRFs within an EDC system. Use of Surrogate Markers (lowering blood pressure, reducing cholesterol) or use of imaging technologies or biochemical biomarkers can help create a more complete picture and provide better investigative data.
1. United States, US Department of Health and Human Services, Food and Drug Administration, Center for Drug Evaluation and Research (CDER) Center for Biologics Evaluation and Research (CBER) Center for Devices and Radiological Health (CDRH),Use of Electronic Health Record Data in Clinical Investigations Guidance for Industry, May 2016.