CONNECT
Adaptive Clinical Systems is an eClinical Technology Solutions provider and “Problem-Solver” providing EDC to CTMS integration and data flow improvements in your clinical trials.
Integrate proprietary and commercial off-the-shelf (COTS) tools and leverage your existing in-house investments, enabling you the freedom to choose the best tools for your study without the need for complex and expensive integration projects. The Adaptive eClinical Bus easily integrates all leading EDC (eg, Medidata, Bioclinica, OpenClinica, etc.), CTMS (eg, IMPACT, Biooptronics, etc.), and Medical Imaging and Analysis tools (eg, Siemens, mint Lesion, etc.).
Simplified Workflow
- Easily track and maintain events and visits in complex studies
- Single-source subject information avoids data duplication
- View study progress in one integrated dashboard
- Tightly integrate patient and investigator reimbursements with eCRF creations in your EDC
Rapid Study Startup
- Quickly identify subjects that meet study inclusion/exclusion criteria
- Directly import demographic information
- Easily map routine visit data into eCRF’s
Seamless Web & Voice Interface
- Reduce transcription errors and avoid complicated data imports/exports
- Easily relay randomization information from IVR/IWRS to EDC/CTMS
- Aggregate ePRO reports as single eCRF in EDC
Reduce Data Management Overhead
- Maintain 21 CFR Part 11 Compliance across all interconnected modules
- Eliminate the need for source data verification
- Streamline risk-based Monitoring and eliminate unnecessary site visits
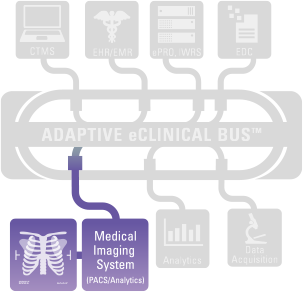
Imaging Integration
- Single source of imaging data
- Native support for both DICOM and proprietary imaging standards
- Support for Criteria ranging from RECIST through mWHO and Cheson

Leverage Real-Time Analytics
- Adaptive eClinical Bus enables continuous updates
- Real-time views of study data
- Easily set up a live link to your sponsor for study data transmissions

Direct Data Capture
- Direct integration with medical device data collectors
- Capture device calibration information in eCRF’s at the time of evaluation
- Seamlessly integrate with lab instruments
