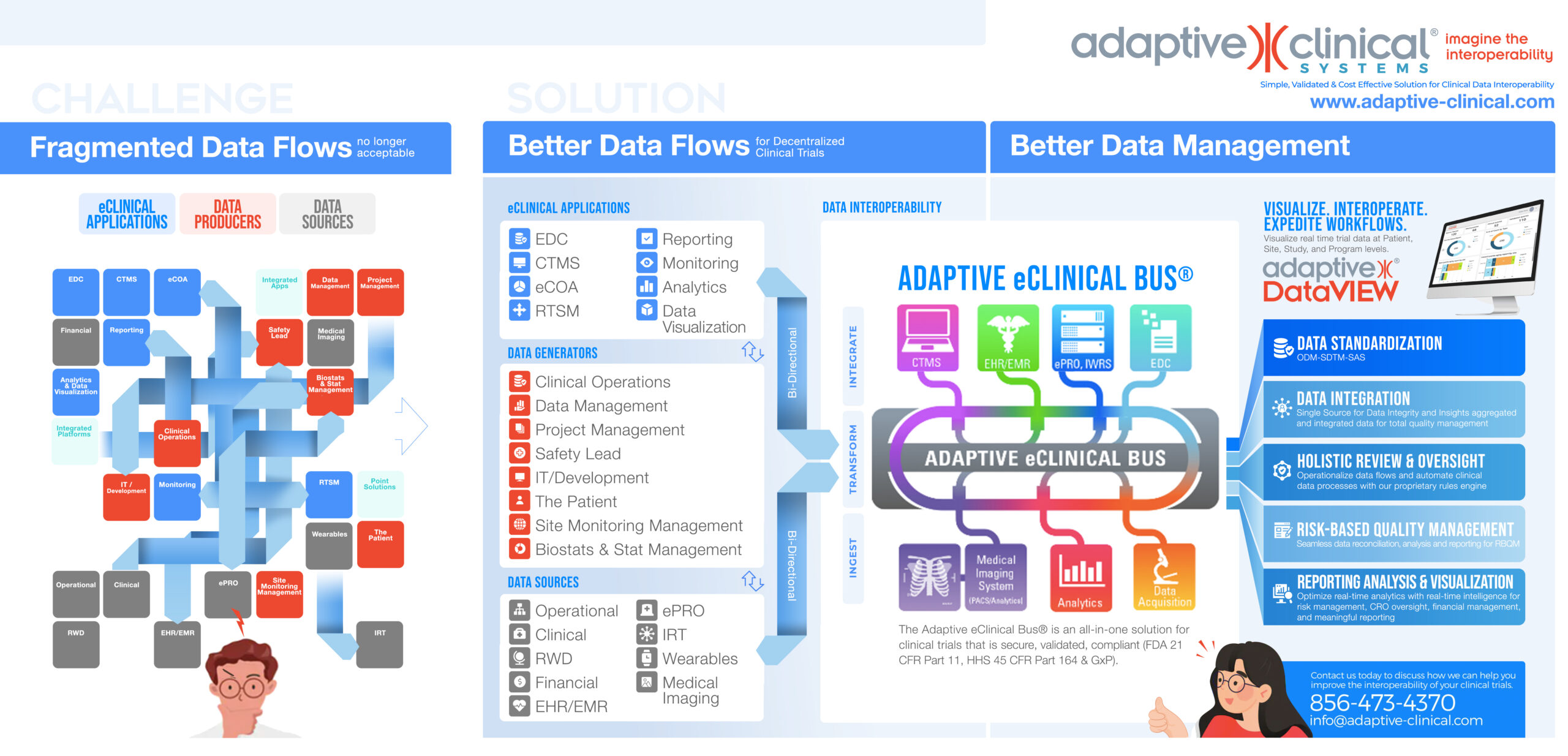
Roadmap to Universal interoperability
Adopt Universal Interoperability for Decentralized Clinical Trials
See the Difference for Better Data Management with Decentralized Clinical Trials and Take the Chaos out of the Increasing Endpoints and eSources. Learn how to create your own Roadmap. Focus on building a best of breed solution with the validated Adaptive eClinical Bus® and achieve universal interoperability. Download today.
Our software helps:

Eliminate duplication of data by capturing and transmitting electronic source data

Encourage entering source data at the point of care and reduce transcription errors

Facilitate remote monitoring of data to reduce the number of onsite visits

Improve site monitoring to minimize the need for cross-reference data in multiple sources

Make it easier for investigators to conduct clinical research

Facilitate the inspection and reconstruction of clinical investigations by FDA
